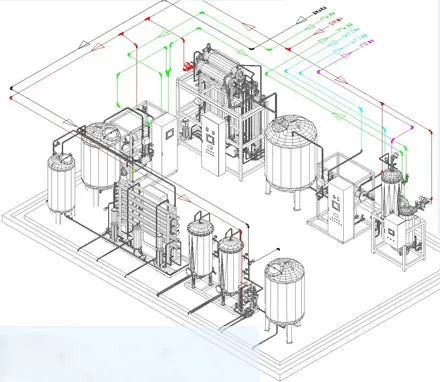
The purified water system is engineered for high-demand standards in electronics, medicine, health products, and food industries. By utilizing filtration, Reverse Osmosis (RO), and sterilization, the raw water is transformed into high-purity water ready for industrial applications. The entire system is constructed from high-grade stainless steel to ensure durability and safety, meeting 2015 Chinese Pharmacopoeia and national GMP standards.

Purified Water Equipment Characteristics
- Fully automatic operation with integrated processing for backwash, regeneration, and disinfection.
- Compliance with GMP requirements using 316L material for back-end equipment and SUS304 for pretreatment.
- Water quality standards: Conductivity ≤0.5μs/cm; TOC ≤200 ppb; Microbial ≤10 cfu/ml.
- Surface treatment with chrome plating for superior rust and corrosion resistance.
- System recovery rates exceeding 80% with concentrated water recovery devices.
Multi-Effect Distilled Water Performance
- Constructed with 316L stainless steel with pickled and passivated working surfaces.
- Uses gas-liquid phase separation to reduce endotoxins by at least 99.99%.
- Special atomizing nozzles ensure even distribution and efficient falling film evaporation.
- Equipped with new expansion connection technology for long-term stable operation.
- Injection water standards: Toxicity 0.25 EU/ml; Microbial ≤10 cfu/100ml.
Water Distribution & Control
- Modular PW/WFI design with compact structure for easy maintenance.
- Circulating pump frequency conversion in accordance with GMP and ISPE guidelines.
- Advanced control system with electronic records, audit tracking, and remote troubleshooting.
- Integrated solutions for purified water, injection water, pure steam, compressed air, and nitrogen.