High-Performance Modular Cleanroom Systems
A clean room is a specially designed environment engineered to exclude airborne particles, bacteria, and other harmful pollutants. It provides precise control over indoor temperature, cleanliness, interior pressure, air velocity, air distribution, noise, vibration, and lighting, including static control within specific demand scopes.
Core Advantage: Regardless of how external air conditions fluctuate, the internal environment maintains its original set requirements for cleanliness, temperature, humidity, and pressure performance characteristics.
The primary function of a clean room is to control the atmospheric cleanliness and humidity for sensitive products (such as silicon or pharmaceuticals) during contact. This specialized space ensures manufacturing occurs in an optimized environment. By international standards, dust-free purification levels are determined by the number of particles per cubic meter exceeding specific diameter standards.
Q1 What core equipment do you manufacture for cleanrooms?
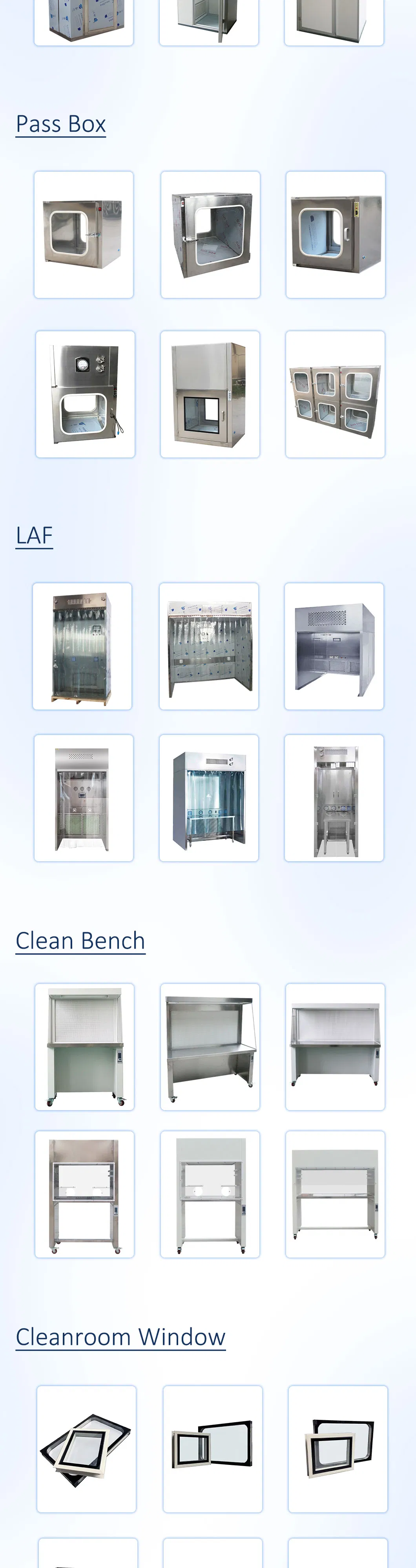
We focus on comprehensive cleanroom projects, including air showers, Fan Filter Units (FFU), pass boxes, dispensing booths, laminar air flow hoods and benches, Air Handling Units (AHU), and hermetic doors & windows.
Q2 What is the standard lead time for orders?
For full cleanroom projects including AHU, the lead time is approximately 35 days. For standard goods without AHU, the lead time is reduced to 10 days.
Q3 What type of warranty and after-sales support is provided?
We provide a 2-year warranty from project completion. During this period, we will replace any faulty components. If technical issues cannot be handled locally, we provide engineering support to resolve them.
Q4 Can you provide cleanroom validation documentation?
Yes, we provide a complete set of documentation including DQ, IQ, OQ, and PQ, as well as FAT & SAT reports, operation manuals, maintenance guidance, and full construction drawings.
Q5 What are your technical advantages in this field?
We have an experienced technical team of 12 senior cleanroom & HVAC engineers. We have successfully completed over 2,100 cleanroom projects globally across various regions including Europe, Southeast Asia, the Middle East, and North America.
Q6 Are the materials used compliant with pharmaceutical standards?
Yes, our systems utilize high-quality Stainless Steel (SUS304/201) and specialized modular designs that meet the stringent hygiene and durability requirements of pharmaceutical and electronic industrial environments.