Frequently Asked Questions
1. What cleanroom classes do you provide?
We offer cleanrooms from ISO Class 5 to Class 8, including GMP A/B/C/D, tailored to pharmaceutical, medical, and industrial applications.
2. Are your cleanrooms compliant with GMP, FDA, and ISO standards?
Yes. Our cleanroom solutions are designed in full compliance with ISO 14644, GMP, FDA, and EU GMP requirements.
3. Can you provide customized cleanroom design based on our process?
Absolutely. Our in-house engineering team provides customized layout and HVAC design based on your process flow, equipment, and capacity needs.
4. How long does cleanroom installation take?
Thanks to our modular cleanroom system, installation typically takes 2–6 weeks, significantly shorter than traditional construction.
5. Can the cleanroom be expanded or modified in the future?
Yes. Our modular structure allows for easy expansion, relocation, or upgrading, reducing future investment risks.
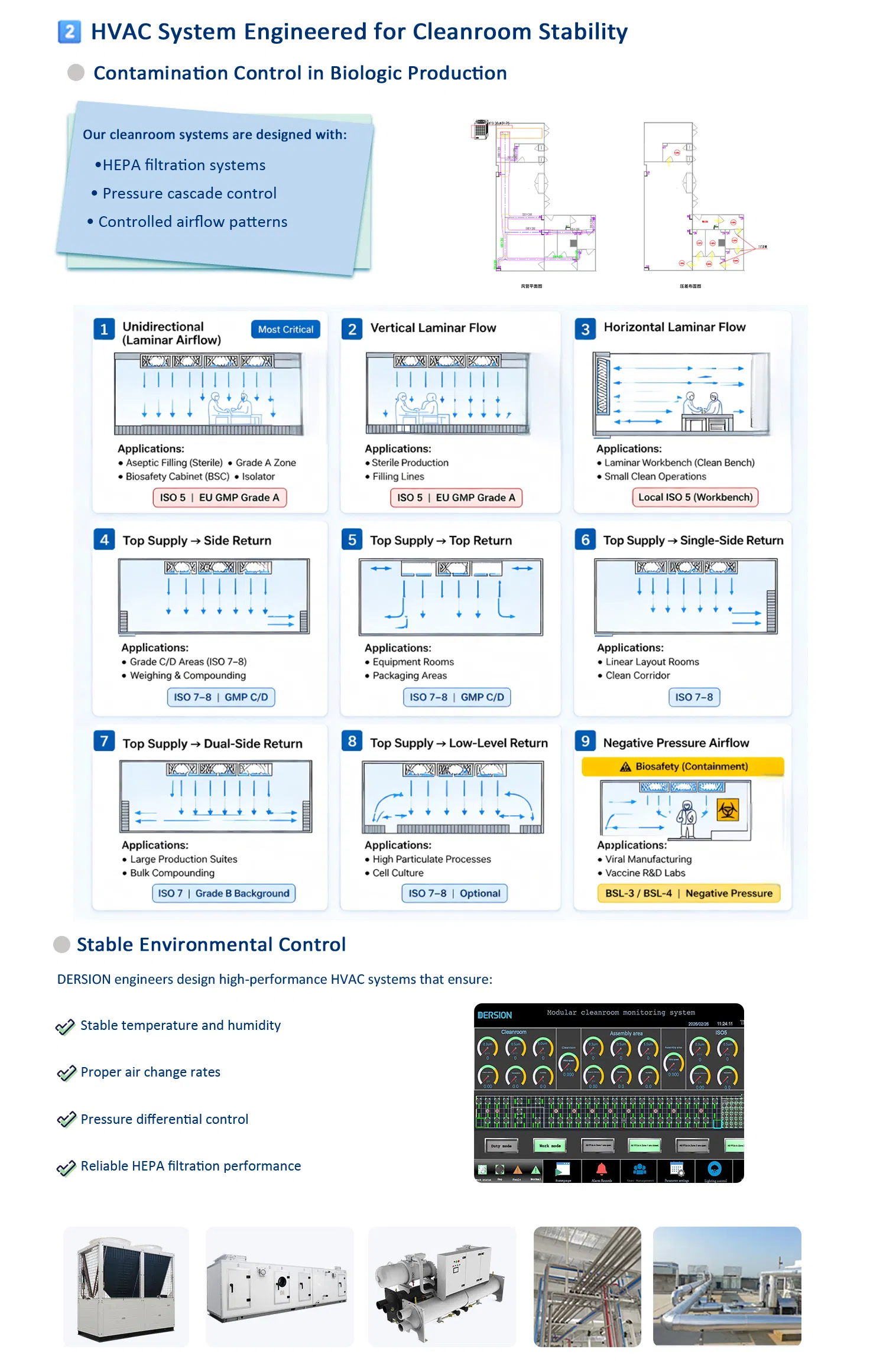
6. How do you control temperature, humidity, and air cleanliness?
We use a professionally engineered HVAC system with HEPA filtration, ensuring stable control of temperature, humidity, pressure, and cleanliness levels.